 
The SARS-CoV-2 S immunogen in the vaccine is expressed in the trimeric pre-fusion conformation the coding sequence has not been modified in order to stabilize the expressed S-protein in the pre-fusion conformation. To be maintained at very low temperaturesĬOVID-19 Vaxzeria (previously COVID-19 vaccine AstraZeneca) is a monovalent vaccine composed of a single recombinant, replication-deficient chimpanzee adenovirus (ChAdOx1) DNA vector encoding the S glycoprotein of SARS-CoV-2 (ChAdOx1-S, AZD1222). Plasmid DNA that contain mammalian expression promotors and the target gene Potential integration of the viral genome into the host genome Replication-incompetent or replication-competent viral vector expressing the target viral protein Reversion to or recombination with the wild-type virus (nucleotide substitution during viral replication)Ĭomposed of viral proteins that have been expressed in one of various systems Stimulate humoral and cellular immunity to multiple components of the whole attenuated virus Genetically weakened versions of the wild-type virus Integrity of the immunogenic particles must be maintained Stable immune response targeting the Spike protein and other components of the virus To this regard, we aimed to summarize main mechanisms of SARS-CoV-2 vaccines and their potential interactions with the cardiovascular system. However, some concerns regarding the safety of SARS-CoV-2 vaccines have been recently raised, mostly based on scattered reports of thromboembolic events, ,. To date, a total of seven SARS-CoV-2 vaccines are available across three different platforms, , and as of 16 April 2021, a total of 734.121.870 vaccine doses have been administered ( ). Nevertheless, vaccines to prevent SARS-CoV-2 infection are considered the most promising approach for curbing the pandemic. Thus, there is an ongoing search for therapeutics finalized to block the transition from infection to severe forms of coronavirus disease 2019 (COVID-19).ĭifferent therapeutic strategies are under scrutiny which include blockade of SARS-CoV-2 from binding to human cell receptors, prevention of the viral ribonucleic acid (RNA) synthesis and replication, the restoration of the host's innate immunity, and the modulation of the host's specific receptors or enzymes. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) rapidly spread across the world and killed more than 2.9 million individuals globally, with 137 million cases being confirmed by laboratory tests (April 16 2021, ). Further research is urgently needed in this area. This might lead to excessively enhanced inflammatory and thrombotic reactions in occasional subjects. 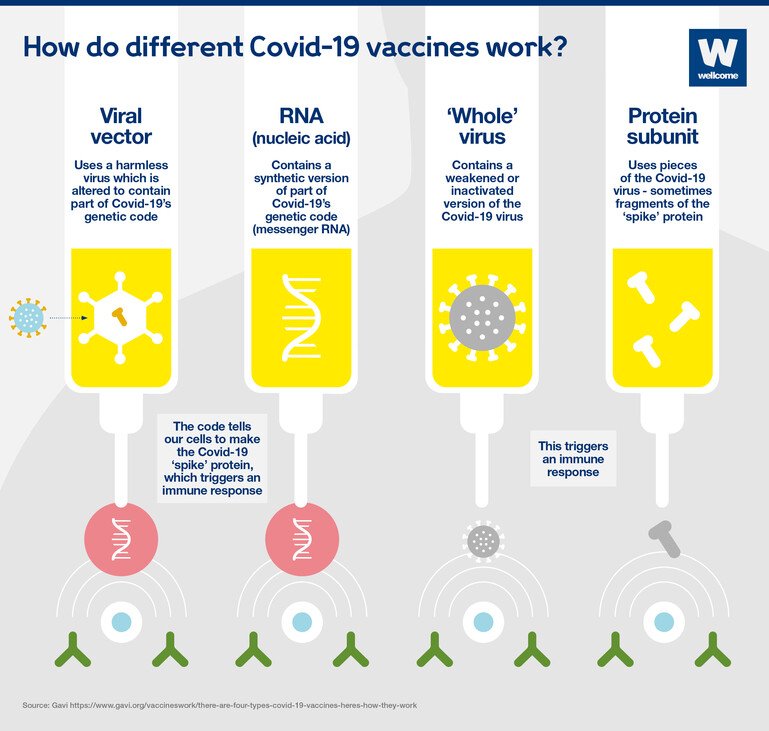
Whereas Phase III vaccine trials generally excluded participants with previous immunization, vaccination of huge populations in the real life will inevitably include individuals with preexisting immunity. These reactions may ultimately lead to platelet aggregation, thrombosis and inflammation mediated by several mechanisms including platelet ACE2 receptors. The free-floating Spike proteins synthetized by cells targeted by vaccine and destroyed by the immune response circulate in the blood and systematically interact with angiotensin converting enzyme 2 (ACE2) receptors expressed by a variety of cells including platelets, thereby promoting ACE2 internalization and degradation. The resulting pathological features may resemble those of active coronavirus disease.  
Such response appears to be quite vigorous in the presence of DNA vaccines which encode viral vectors, as well as in subjects who are immunized because of previous exposure to SARS-CoV-2. These proteins are recognized by the immune system which rapidly develops an immune response. Once synthetized, the Spike proteins assembled in the cytoplasma migrate to the cell surface and protrude with a native-like conformation. Vaccines increase the endogenous synthesis of SARS-CoV-2 Spike proteins from a variety of cells. Different vaccine platforms are currently available which include live attenuated vaccines, inactivated vaccines, recombinant protein vaccines, vector vaccines, DNA vaccines and RNA vaccines. Some mechanisms have been suggested which might explain the adverse cardiovascular reactions to SARS-CoV-2 vaccines. Some concerns have been raised regarding the safety of SARS-CoV-2 vaccines, largely based on case-reports of serious thromboembolic events after vaccination. Vaccines to prevent acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection elicit an immune neutralizing response. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
